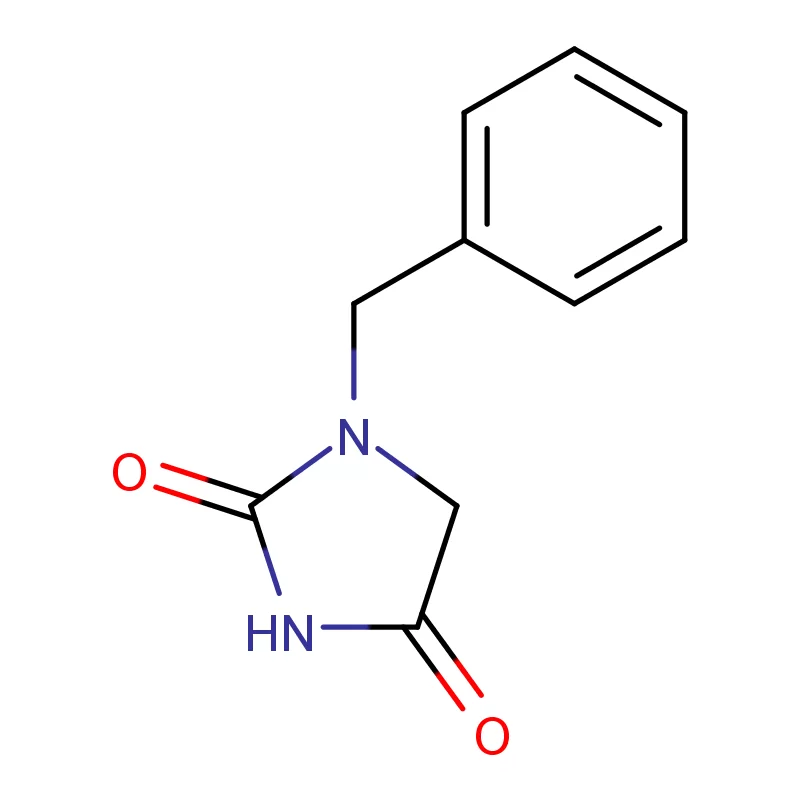
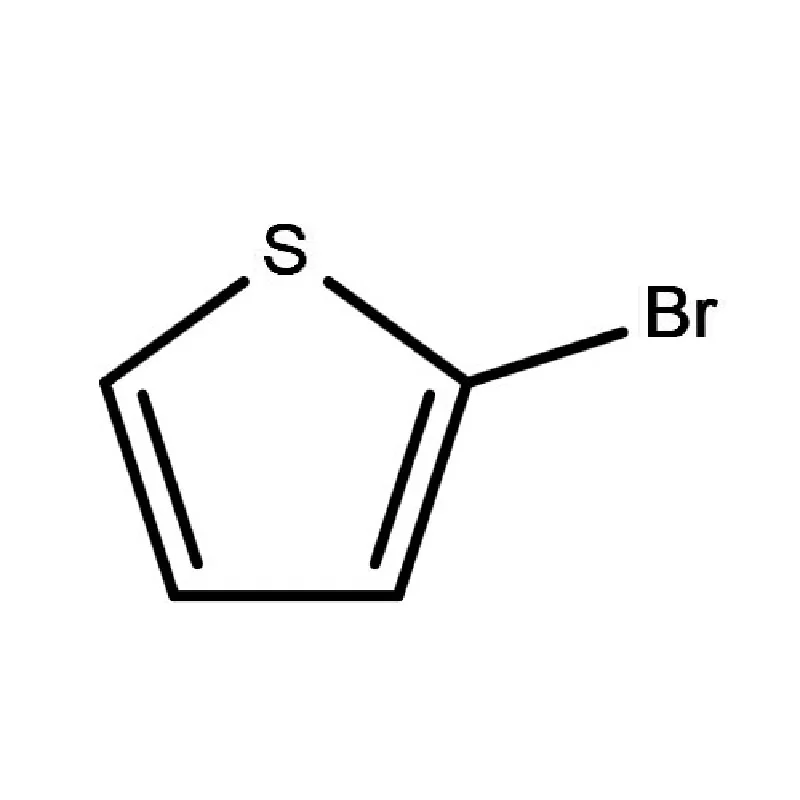
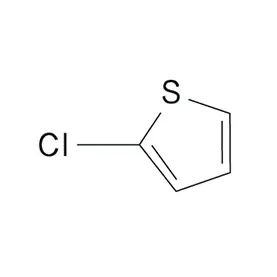
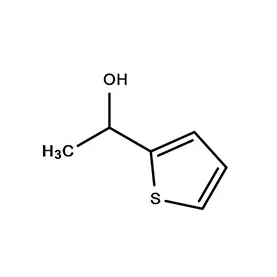
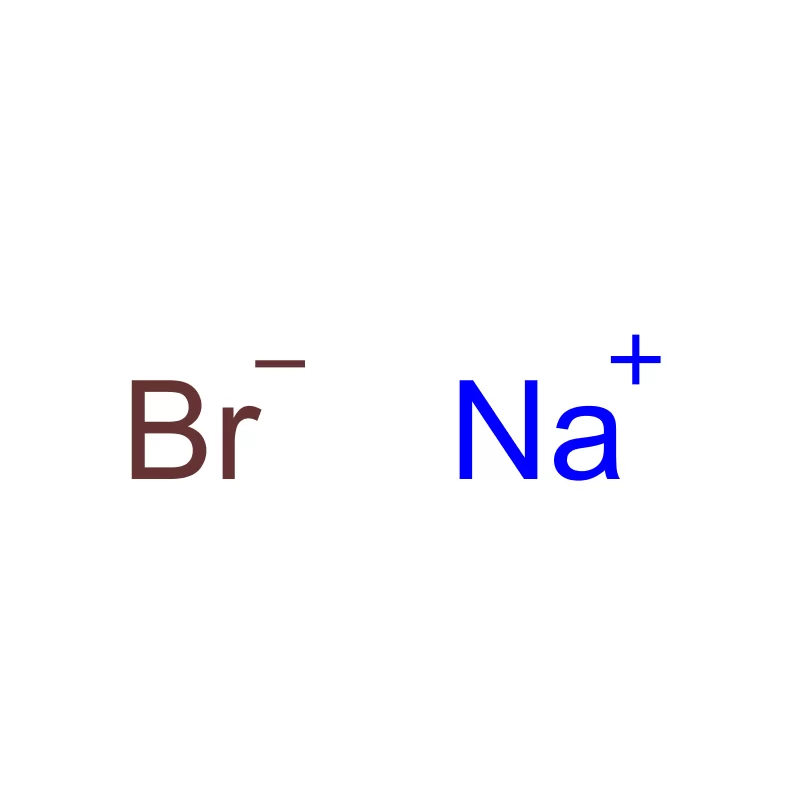Japan presents a unique market for pharmaceutical intermediates, characterized by stringent quality control, a highly regulated environment, and a strong emphasis on innovation. The country’s aging population drives consistent demand for advanced pharmaceutical products. The geographical challenges of Japan, including frequent seismic activity, necessitate robust supply chain management.
The demand for fine chemicals is continuously increasing in Japan due to the expansion of the specialty chemicals sector and the growing need for high-purity compounds in pharmaceutical and electronic applications. A significant portion of production is concentrated around major industrial hubs like Tokyo, Osaka and Nagoya.
Japanese manufacturers prioritize sustainability and eco-friendly processes, creating opportunities for suppliers offering green chemistry solutions, including efficient industrial water treatment chemicals. The market requires consistent quality, reliable supply, and adherence to strict regulatory standards.