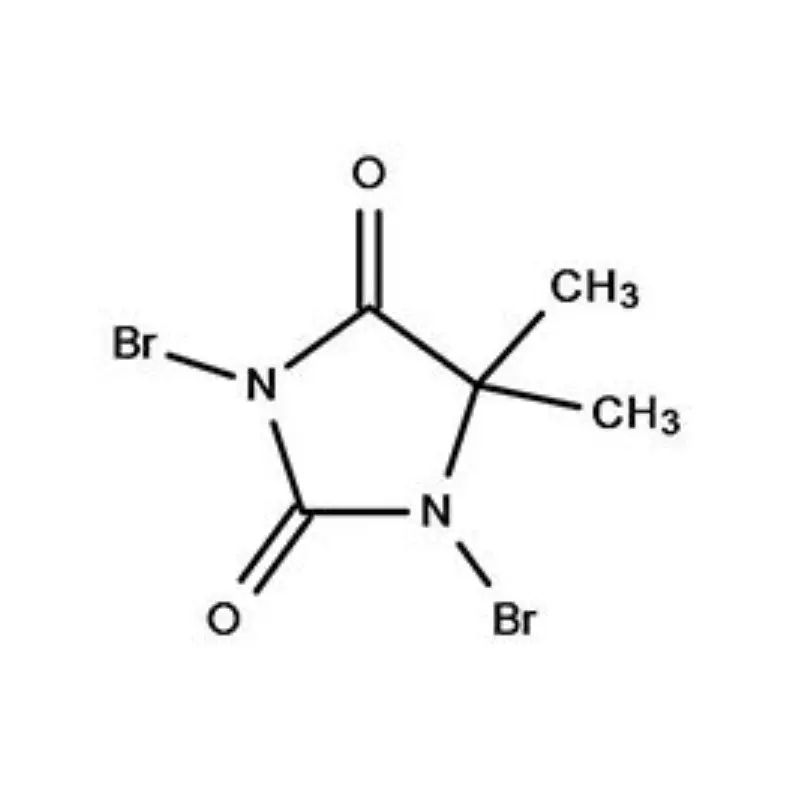
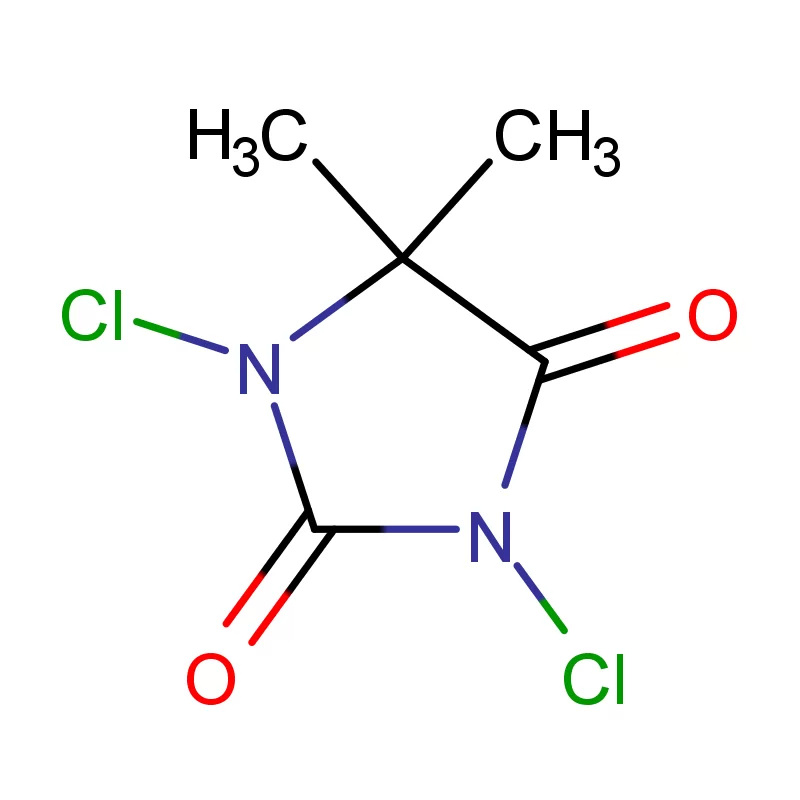
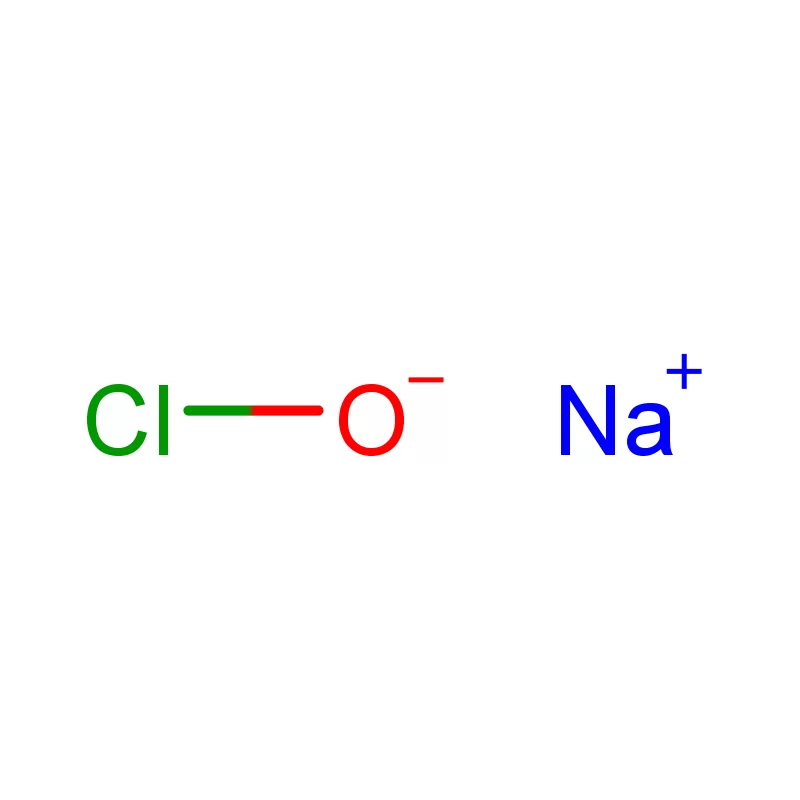
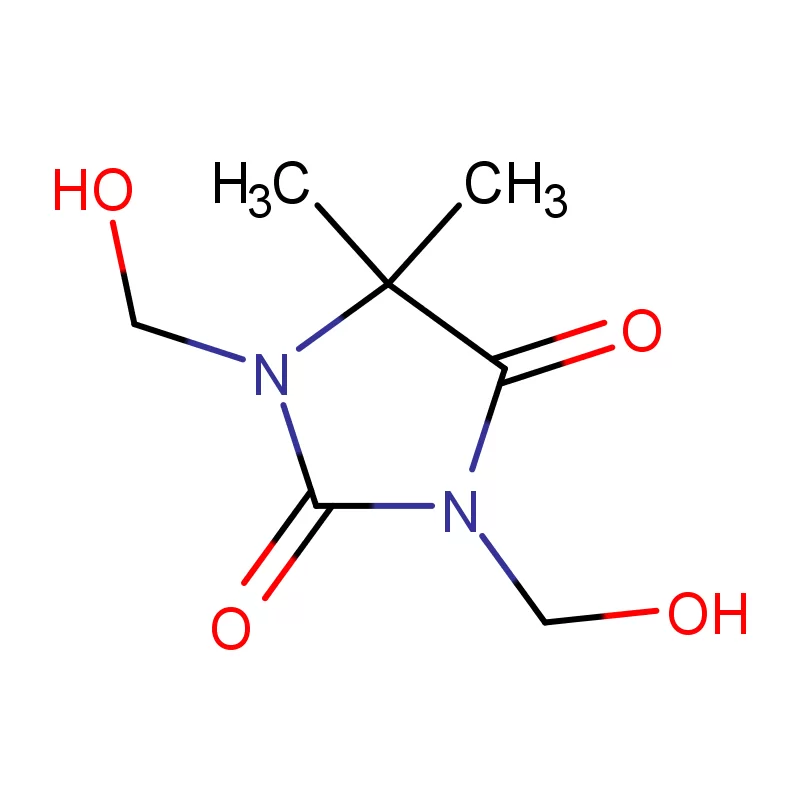
Tajikistan, a landlocked country in Central Asia, presents a unique set of conditions for the pharmaceutical intermediates industry. Its mountainous terrain and limited infrastructure pose logistical challenges, while a growing population and increasing healthcare demands drive a need for locally sourced pharmaceutical products. The industry is still developing, with a reliance on imports for many key raw materials.
The economic climate in Tajikistan is characterized by a transition towards a market economy, with the pharmaceutical sector receiving increased attention from both government and private investors. Demand for fine chemicals is rising alongside the expansion of local pharmaceutical manufacturers. Access to consistent quality supplies remains a significant hurdle for many companies.
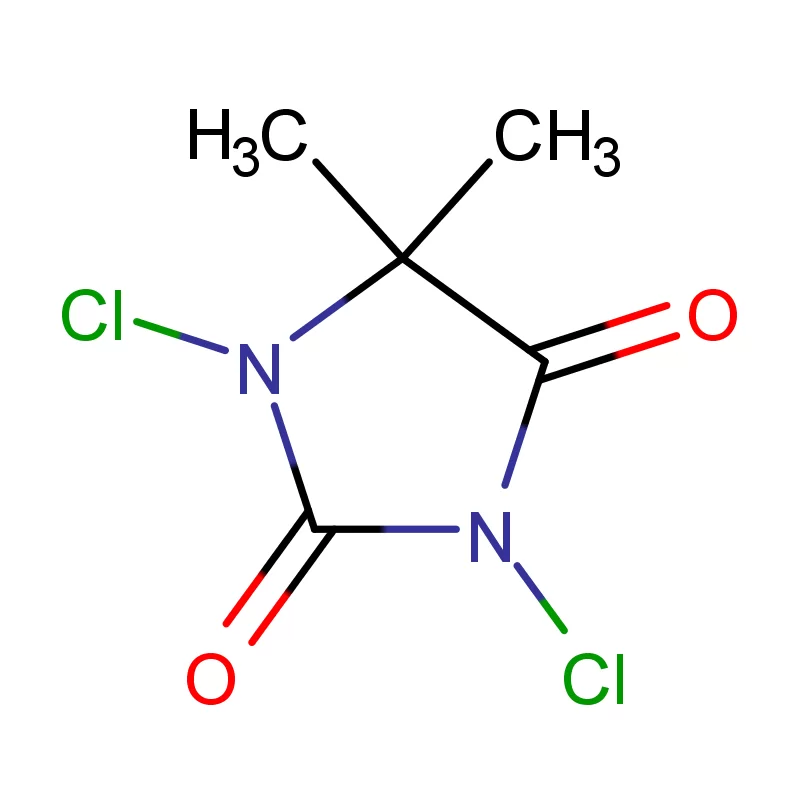
The regulatory landscape for pharmaceuticals is evolving, with efforts to align with international standards. This creates opportunities for companies offering high-quality, compliant 2 Bromothiophene and other essential chemical building blocks. Navigating these regulations effectively is crucial for success in this market.